 
Textbook content produced by OpenStax is licensed under a Creative Commons Attribution License. We recommend using aĪuthors: Paul Flowers, Klaus Theopold, Richard Langley, William R. Use the information below to generate a citation. 
Then you must include on every digital page view the following attribution: If you are redistributing all or part of this book in a digital format, Then you must include on every physical page the following attribution: If you are redistributing all or part of this book in a print format, Want to cite, share, or modify this book? This book uses the If the molecular (or molar) mass of the substance is known, it may be divided by the empirical formula mass to yield the number of empirical formula units per molecule ( n): As the name suggests, an empirical formula mass is the sum of the average atomic masses of all the atoms represented in an empirical formula. Molecular formulas are derived by comparing the compound’s molecular or molar mass to its empirical formula mass. Molar mass can be measured by a number of experimental methods, many of which will be introduced in later chapters of this text. Molecular mass, for example, is often derived from the mass spectrum of the compound (see discussion of this technique in the previous chapter on atoms and molecules). These quantities may be determined experimentally by various measurement techniques. Determining the absolute numbers of atoms that compose a single molecule of a covalent compound requires knowledge of both its empirical formula and its molecular mass or molar mass. 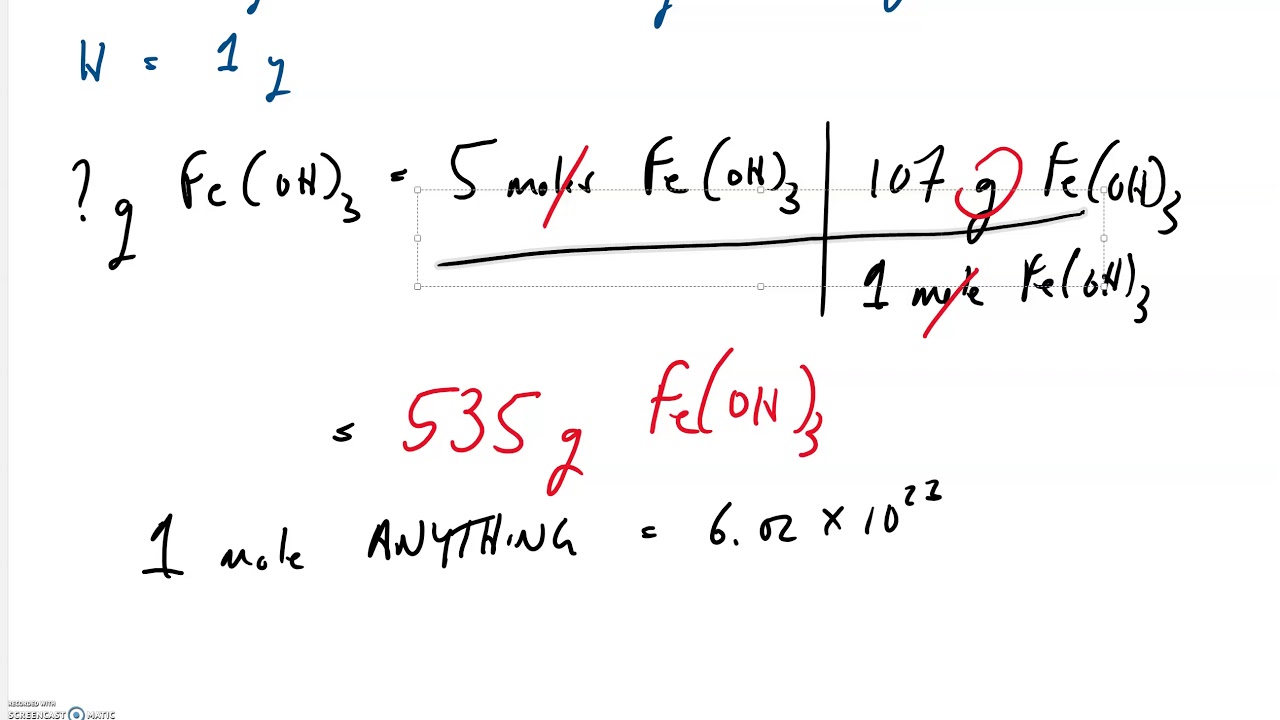
Recall that empirical formulas are symbols representing the relative numbers of a compound’s elements. The percent composition of this compound could be represented as follows: For example, consider a gaseous compound composed solely of carbon and hydrogen. The results of these measurements permit the calculation of the compound’s percent composition, defined as the percentage by mass of each element in the compound. When a compound’s formula is unknown, measuring the mass of each of its constituent elements is often the first step in the process of determining the formula experimentally. Sometimes when the volume of a component is specified, in order to get the mass of. The elemental makeup of a compound defines its chemical identity, and chemical formulas are the most succinct way of representing this elemental makeup. Molarity is defined as Moles of a component divided my volume of solvent. So, for a value ‘x’, its percentile can be expressed as the ratio of the number of denominations below ‘x’ to the total number of denominations. But what if the chemical formula of a substance is unknown? In this section, these same principles will be applied to derive the chemical formulas of unknown substances from experimental mass measurements. The percentile formula is also used to calculate income, weight, etc. Given the chemical formula of the substance, one may determine the amount of the substance (moles) from its mass, and vice versa. The previous section discussed the relationship between the bulk mass of a substance and the number of atoms or molecules it contains (moles). How to Calculate Percent Recovery Data given: Amount of zinc (original) 11.23 gm Solution: Percent recovery (amount of substance recovered on purification. Determine the molecular formula of a compound.Determine the empirical formula of a compound.Compute the percent composition of a compound.Following are the frequently asked questions on the mass percent. You can use this knowledge to solve numerical problems involving solution concentration. That way, the reader can critically evaluate the meaning of the data themselves.By the end of this section, you will be able to: The article Mass Percent Formula will assist you in understanding mass by mass percentage formula, mass by volume percentage, and volume by volume percentage. Empirical Formula Rhyme (to help you remember the steps). I would say depending on whether the errors on the molar mass determination or the elemental composition are larger, you would have a different top candidate, and should report the numerically exact formula with non-integer coefficients. knowing the mass of each element present in the whole compound. $$ n = 67.3 \% \cdot \frac$ is a good answer, but the data are not consistent, so it is not the complete answer. That way, you can directly calculate how many moles of each element are in one mole of compound: Rather than starting out with 100 g of unknown, I would start out with 1 mol of unknown, i.e. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
